

By signing in or creating an account, you agree with Associated Broadcasting Company's Terms & Conditions and Privacy Policy.


By signing in or creating an account, you agree with Associated Broadcasting Company's Terms & Conditions and Privacy Policy.
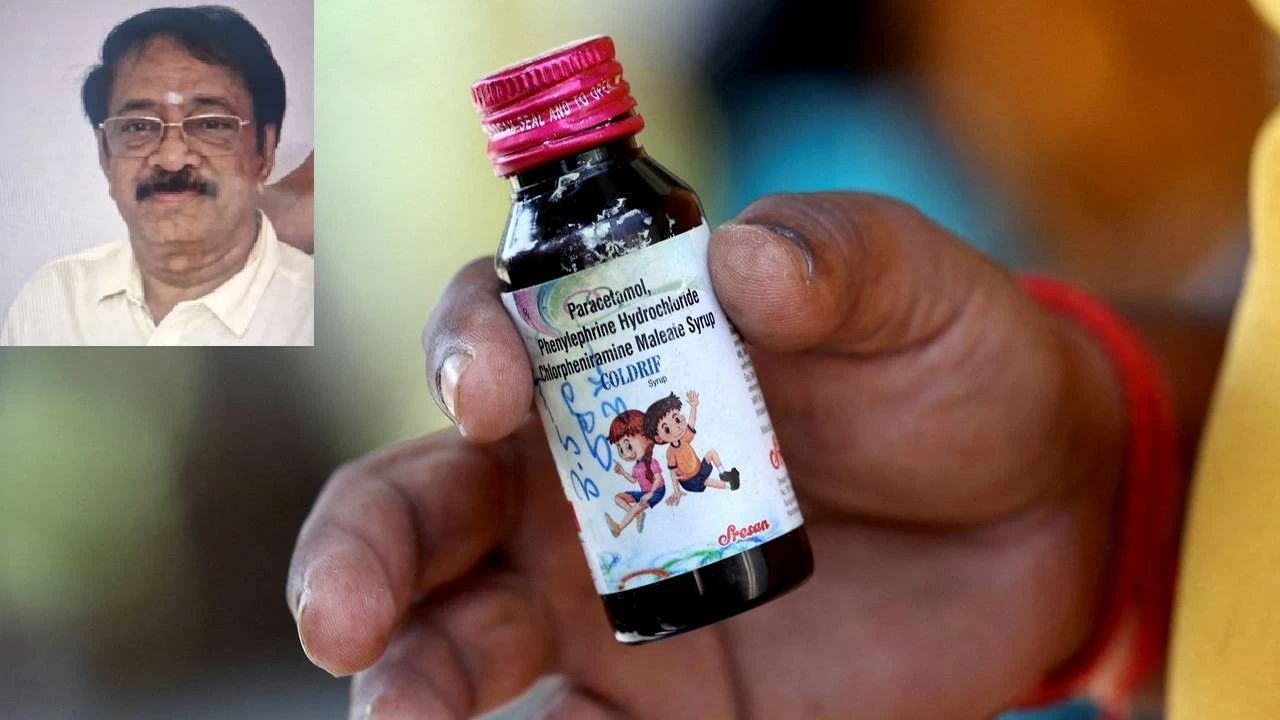
Chennai: Amid a massive row over deaths of more than 20 children in several states, mostly in Madhya Pradesh, allegedly due to contaminated cough syrup Coldrif, the owner of the Tamil Nadu-based pharmaceutical firm -- Sresan Pharmaceuticals -- was arrested late Wednesday night. S Ranganathan Govindan, the owner of the pharma firm that manufactured Coldrif, was taken into custody from Chennai by a team of police from Madhya Pradesh.
Chhindwara Superintendent of Police (SP) Ajay Panday confirmed that the Special Investigation Team (SIT) from Madhya Pradesh, arrested Govindan around 2 am. He will be presented before Chennai court and brought to Chhindwara in MP after securing his transit remand, the SP said. The police had announced a reward of Rs 20,000 for information leading to his arrest.
Coldrif, a cough syrup manufactured by Sresan Pharmaceuticals in Tamil Nadu's Kancheepuram, has been linked to the deaths of more than 20 children in Madhya Pradesh and Rajasthan. Laboratory tests revealed that the cough syrup contained diethylene glycol -- a highly toxic industrial solvent prohibited for pharmaceutical use. It is suspected that the toxic chemical severely damaged the kidneys of the children.
What actions taken so far over cough syrup tragedy
Earlier, Tamil Nadu government cancelled the licence of Sresan Pharmaceutica, after several children mostly below the age of 15 died due to kidney failure after allegedly consuming the contaminated cough syrup.
Amid immense outrage over the children's deaths, several states, including Madhya Pradesh, Tamil Nadu, Rajasthan, Maharashtra, Punjab, Goa, Haryana, Himachal Pradesh, Karnataka, Uttar Pradesh, and Uttarakhand, have banned the sale and consumption of the cough syrup.
Madhya Pradesh Police had last week arrested Praveen Soni, a senior paediatrician, for alleged negligence. Soni, a government doctor who prescribed the syrup to children after they visited his private clinic in Madhya Pradesh, was also suspended.
Doctors' body seeks probe into cough syrup deaths
On Wednesday, the Federation of All India Medical Association (FAIMA) on Wednesday wrote to the Ministry of Health urging it to set up a central committee to probe the cases. "The incident has raised serious questions about the safety, quality assurance, and monitoring mechanisms governing the manufacture and distribution of pharmaceutical products for children," the doctors' body said.
FAIMA called for a "nationwide inspections and random testing" of paediatric medicines and syrups to verify compliance with established quality standards.












